NSYSU team developed a 15-minute COVID-19 test to detect virus even in an early stage of infection


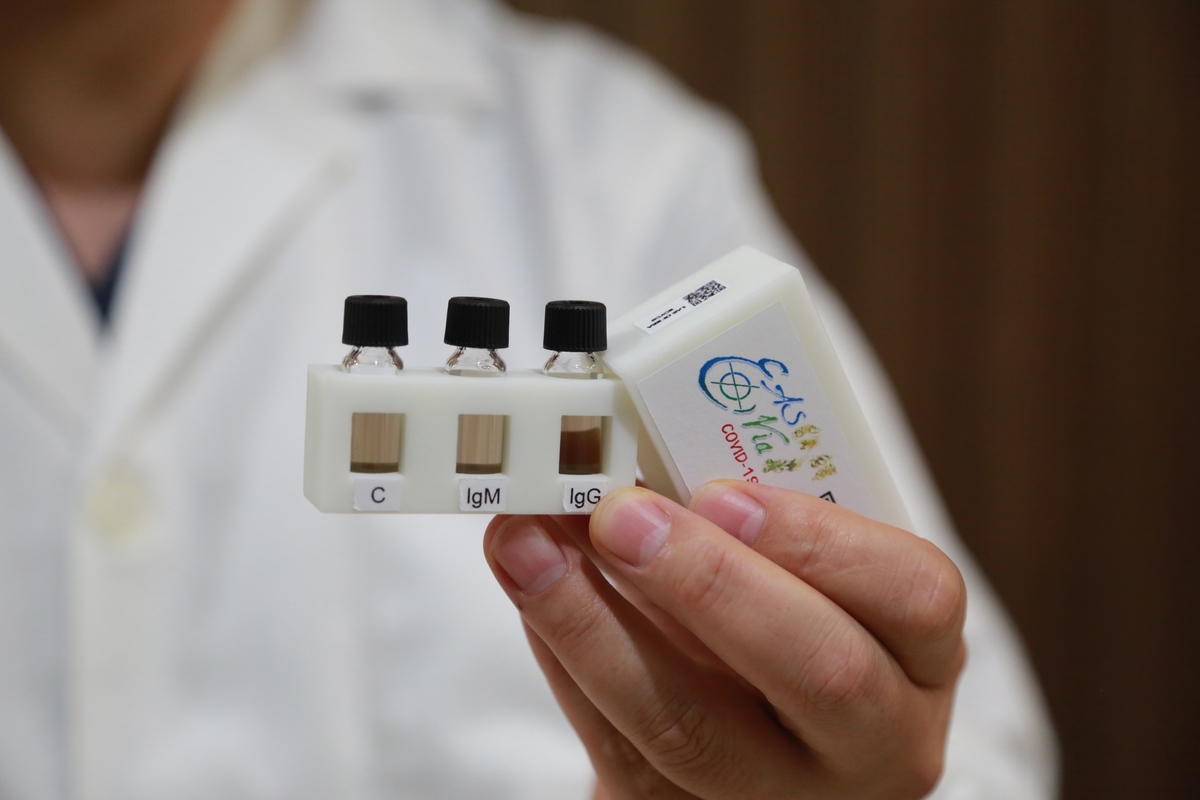


One drop of blood is enough to detect COVID-19 even in patients in an early stage of infection without clear symptoms in just 15 minutes! The research team of Associate Professor Hung-Wei Yang of the Institute of Medical Science and Technology of National Sun Yat-sen University developed a rapid test kit using a highly catalytic nanomaterial and biomolecular patterning techniques to create an original ultra-sensitive measurement flask (EasyVial). It is a different technology than the one applied by the tests available on the market, which are based on the same technology as home pregnancy tests. The new technology developed by NSYSU greatly reduces the testing time that the traditional enzyme-linked immunosorbent assay (ELISA) would require. No medical personnel is needed in the process: a drop of blood is enough to detect IgM antibodies in a patient in an early stage of COVID-19 infection showing no symptoms in just 15 minutes.
While the COVID-19 pandemic is spreading around the world, virus tests are scrutinized for accuracy. False-negative and false-positive results were obtained not only in China; the case of a 17-year-old infected patient tested negative 12 times in a row in South Korea spurred a debate on the accuracy of virus test kits. Member of the Institute of Medical Science and Technology and Rapid Screening Research Center for Toxicology and Biomedicine, NSYSU, Associate Professor Hung-Wei Yang pointed out that most of the currently available clinical rapid tests can detect an early infection using a rapid-testing strip (applied in home pregnancy tests) that has an accuracy of 65 ng/mL and thus, “the probability of false-negative result is very high in case of an early stage of infection”. The test developed by the NSYSU team is not only over 6,000 times more accurate – 0.01 ng/mL but can be done at home.
Associate Professor Hung-Wei Yang led three doctoral students: Ying-Pei Hsu of the Department of Materials and Optoelectronic Science, Nan-Hsi Li and Hao-Han Pang of the Institute of Biomedical Sciences to form a research team and use their previous experience in the development of rapid tests for early detection of cancer to create an original ultra-sensitive measurement flask (EasyVial) and conduct verification by clinical blind test in collaboration with the team of Associate Professor Sheng-Fan Wang of the Department of Medical Laboratory Science and Biotechnology, Kaohsiung Medical University (KMU). The research team conducted a blind test on a patient who returned to Taiwan from abroad and was tested positive by an RT-PCR nucleic acid detection kit, however, it showed no symptoms of the disease. They isolated patient’s antibodies by suction for verification. In the blood sample taken from the patient on the first day of the patient’s stay at the hospital, they could precisely detect traces of novel coronavirus IgM antibodies in an early stage of infection (the test allows the detection of IgM antibodies even with 10,000 times smaller concentration). Still, an early-stage infection could be proved without IgM antibodies detected, which corresponds to the result of clinical tests. At the same time, they conducted a test on the blood sample using the traditional ELISA kit; it detected no concentration of antibodies (negative result). On the third day, they could detect IgM and IgM antibodies in the blood sample, proving the tremendous efficacy of the ultra-sensitive measurement flask in the rapid detection of the virus even in an early stage of infection when the patient shows no symptoms.
“Fast detection at an early stage of infection when there are no symptoms of COVID-19 allows early isolation and treatment of the patient and prevents possible transmission of the virus”. Associate Professor Hung-Wei Yang emphasized that currently if COVID-19 is detected in an initial test, further confirmation is needed with an RT-PCR nucleic acid test, which not only requires the participation of medical personnel, who incur a risk of infection when collecting samples but also, the result can be obtained only after four hours, and sometimes multiple testing is required. The ultra-sensitive measurement flask (EasyVial) can be further applied to detect communicable diseases, caused by such viruses as Zika, dengue, and Ebola. The test can come in handy during virus spreading seasons or in case of an epidemic outbreak, greatly reducing the engagement of medical personnel and preventing the spread of the epidemic.
While the COVID-19 pandemic is spreading around the world, virus tests are scrutinized for accuracy. False-negative and false-positive results were obtained not only in China; the case of a 17-year-old infected patient tested negative 12 times in a row in South Korea spurred a debate on the accuracy of virus test kits. Member of the Institute of Medical Science and Technology and Rapid Screening Research Center for Toxicology and Biomedicine, NSYSU, Associate Professor Hung-Wei Yang pointed out that most of the currently available clinical rapid tests can detect an early infection using a rapid-testing strip (applied in home pregnancy tests) that has an accuracy of 65 ng/mL and thus, “the probability of false-negative result is very high in case of an early stage of infection”. The test developed by the NSYSU team is not only over 6,000 times more accurate – 0.01 ng/mL but can be done at home.
Associate Professor Hung-Wei Yang led three doctoral students: Ying-Pei Hsu of the Department of Materials and Optoelectronic Science, Nan-Hsi Li and Hao-Han Pang of the Institute of Biomedical Sciences to form a research team and use their previous experience in the development of rapid tests for early detection of cancer to create an original ultra-sensitive measurement flask (EasyVial) and conduct verification by clinical blind test in collaboration with the team of Associate Professor Sheng-Fan Wang of the Department of Medical Laboratory Science and Biotechnology, Kaohsiung Medical University (KMU). The research team conducted a blind test on a patient who returned to Taiwan from abroad and was tested positive by an RT-PCR nucleic acid detection kit, however, it showed no symptoms of the disease. They isolated patient’s antibodies by suction for verification. In the blood sample taken from the patient on the first day of the patient’s stay at the hospital, they could precisely detect traces of novel coronavirus IgM antibodies in an early stage of infection (the test allows the detection of IgM antibodies even with 10,000 times smaller concentration). Still, an early-stage infection could be proved without IgM antibodies detected, which corresponds to the result of clinical tests. At the same time, they conducted a test on the blood sample using the traditional ELISA kit; it detected no concentration of antibodies (negative result). On the third day, they could detect IgM and IgM antibodies in the blood sample, proving the tremendous efficacy of the ultra-sensitive measurement flask in the rapid detection of the virus even in an early stage of infection when the patient shows no symptoms.
“Fast detection at an early stage of infection when there are no symptoms of COVID-19 allows early isolation and treatment of the patient and prevents possible transmission of the virus”. Associate Professor Hung-Wei Yang emphasized that currently if COVID-19 is detected in an initial test, further confirmation is needed with an RT-PCR nucleic acid test, which not only requires the participation of medical personnel, who incur a risk of infection when collecting samples but also, the result can be obtained only after four hours, and sometimes multiple testing is required. The ultra-sensitive measurement flask (EasyVial) can be further applied to detect communicable diseases, caused by such viruses as Zika, dengue, and Ebola. The test can come in handy during virus spreading seasons or in case of an epidemic outbreak, greatly reducing the engagement of medical personnel and preventing the spread of the epidemic.
Click Num:
Share
