Solar fuels circulating carbon dioxide likely to become a solution to climate change


2021-07-08
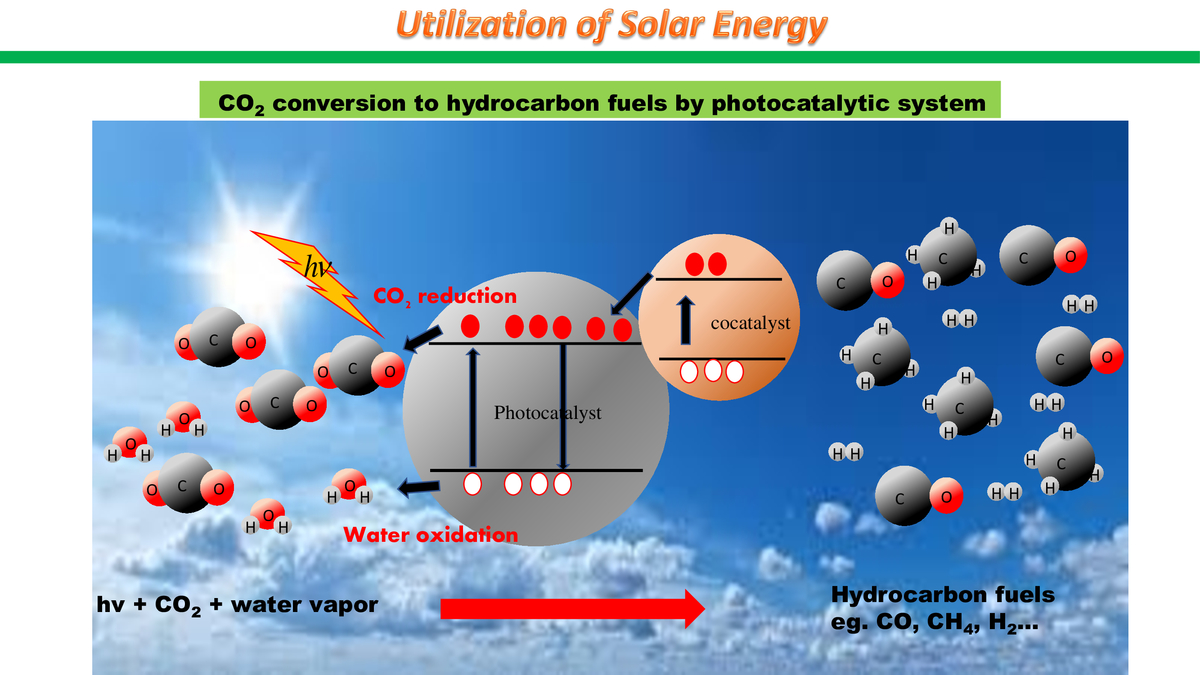
Solar energy is an abundant, clean, and renewable source, however, it has been greatly underutilized. According to a report by World Energy Council, “the total annual solar radiation falling on the Earth is more than 7 500 times the world’s total annual primary energy consumption of 450 EJ.” What many scientists are racking their brains on now, is the development of optimized photocatalysts for CO2 conversion to produce storable solar fuels and recycle immediate CO2 emissions. This could become an alternative to fossil fuels and batteries, help solve the problem of energy shortage and reduce global CO2 emissions, which surpass 30 gigatons annually every year of the past decade, according to Global Carbon Project.
Light-to-fuel photocatalytic systems and solar cells (or panels), produce different outcomes: solar cells generate electricity for immediate use depending on the availability of sunlight, while photocatalytic systems consume CO2 to convert it into hydrocarbon fuels, such as methane or ethane, that can be stored for later use. When burned, these fuels release CO2, which, connected to photocatalysts again, can be recycled and circulate. In the future, solar fuels can be an eco-friendly energy solution for factories that emit CO2 in large amounts.
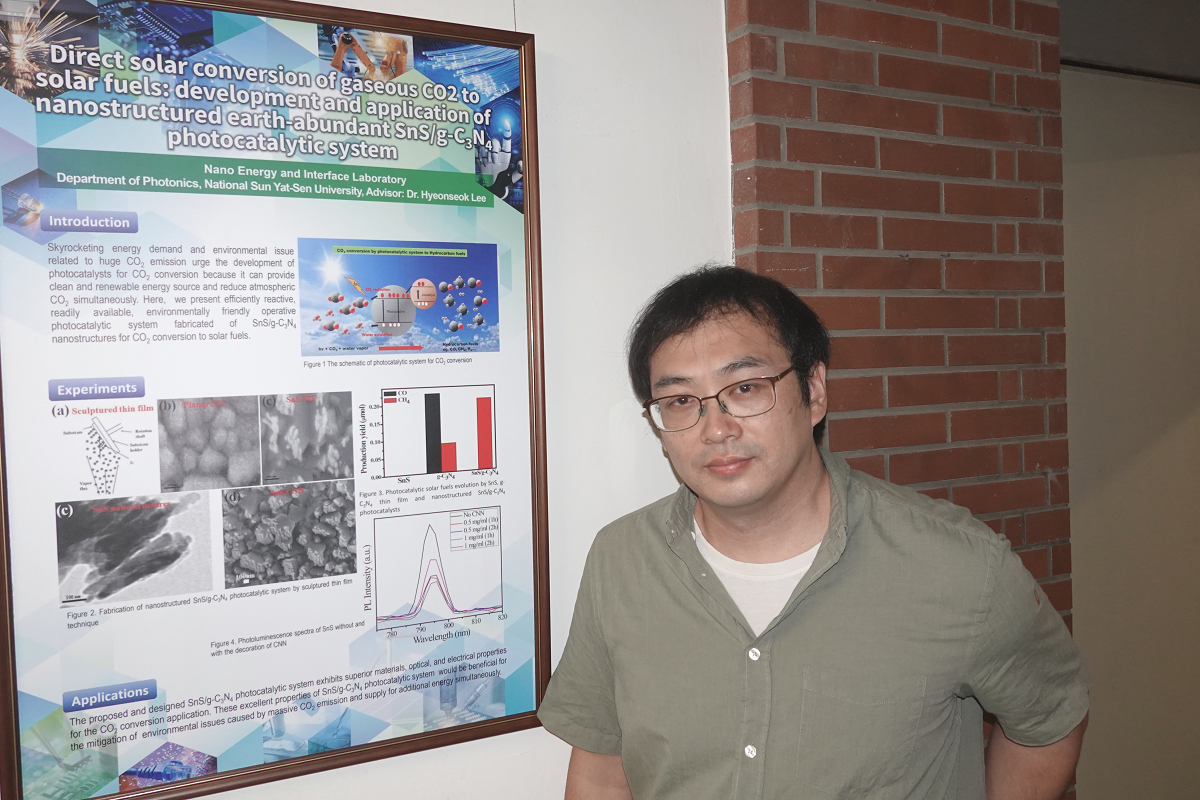
“This research is still in a very initial stage,” says Assistant Professor Hyeonseok Lee of the Department of Photonics, NSYSU, who works on optimized nanostructured photocatalysts to efficiently and selectively generate hydrocarbon solar fuels with long-term stability. With a recently published research paper, Professor Lee and his international research team were the first to demonstrate TiO2 nanotube (TNT) photocatalysts equipped with two different perovskite quantum dots (PQDs) based on methylammonium halides and investigate their fundamental properties and discover their potential for more efficient CO2 conversion, proving the obtained material’s ability to absorb a wider range of light energy and a superior charge transport than other materials studied up to date.
“Nanomaterials have a lot of exposed surface and thus a lot of reaction site, which is very important for photocatalysts. We can engineer their electrical or optical properties, depending on physical parameters such as size or shape, for a more stable environment, a higher efficiency, or highly selective production of hydrocarbon fuels. What’s more, one-dimensional nanomaterials, such as nanotubes or nanowires, promise a much more efficient CO2 conversion as they have special electrical properties.” PQDs, already well known for solar cells, increase chances of utilizing solar energy and extending available wavelengths to visible light.
However, the research on light-to-fuel production by photocatalytic systems is still in its initial stage as at the time being, these systems’ conversion efficiency is much lower than in solar cells, says Professor Lee. Another issue is to produce a reusable material with long-term stability that would be valuable for commercialization. Nonetheless, Lee is optimistic about the future of solar fuels generation. “If we find the proper application, we could use the technology right away. It’s a matter of ideas.”
Solar energy can pave the way to achieve global energy security and environmental justice. All countries, regardless of levels of economic development experience shortage of energy due to environmental issues, says Professor Lee. “My belief is that solar energy can be a solution for all the countries on Earth fairly and evenly regardless of their economic levels. Solar energy doesn’t require a big infrastructure and supplies energy without air pollution. For example, if African countries, with their enormous solar resources, develop solar farms and solar energy conversion systems, they can entirely satisfy their energy demands.”
Read more:
Low-Dimensional Nanostructured Photocatalysts for Efficient CO2 Conversion into Solar Fuels
https://doi.org/10.3390/catal11040418
Toward efficient photocatalysts for light-driven CO2 reduction: TiO2 nanostructures decorated with perovskite quantum dots
https://iopscience.iop.org/article/10.1088/2632-959X/abf3d2/pdf
