Emulating photosynthesis: NSYSU develops new photocatalytic structure to convert carbon dioxide into fuel under bifacial light illumination




2026-03-19
If humanity could emulate plants by using sunlight to convert carbon dioxide (CO2) into energy, a new solution for global warming would be within reach. A research team led by Associate Professor Hyeonseok Lee from the Department of Photonics at National Sun Yat-sen University (NSYSU) has successfully emulated the principles of photosynthesis to develop a novel nanostructured photocatalyst. This material utilizes solar energy to convert CO2 into fuels such as methane (CH4), achieving the dual benefits of reducing greenhouse gases and generating renewable energy. This innovative research has been published in the prestigious international journal, Applied Catalysis B: Environment and Energy.
In nature, plants perform photosynthesis daily by absorbing sunlight to convert CO2 and water into nutrients while releasing oxygen, acting as natural "solar factories." Scientists are now attempting to integrate this capability into the technological world. Hyeonseok Lee noted that as global warming and climate change intensify, the scientific community has been eager to develop "artificial photosynthesis" technology to use sunlight to convert CO2 back into energy. However, existing photocatalytic technologies face several bottlenecks, such as limited light absorption ranges and rapid recombination of electrons, resulting in low reaction efficiency and difficulty in stably producing specific fuels.
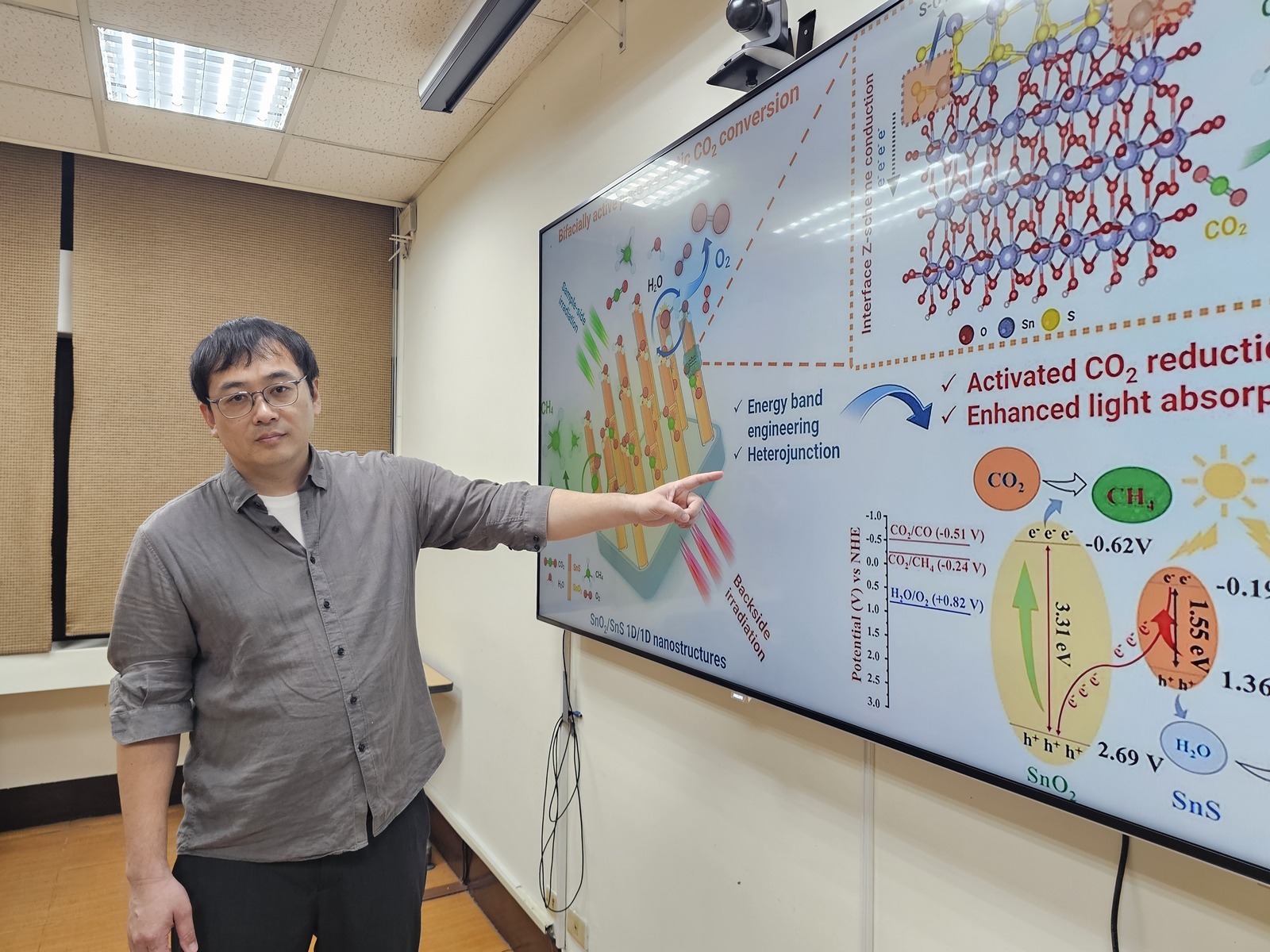
To overcome these limitations, the NSYSU research team designed a one-dimensional (1D/1D) nanocomposite structure composed of tin dioxide (SnO2) and tin sulfide (SnS). By vertically growing SnS nanorods on the surface of SnO2 nanocolumns, they created a sharper-shaped hetero-nanostructure. This design not only increases the surface area but also allows light to be captured from various angles, significantly enhancing light utilization efficiency. The team pointed out that because the sun's position changes throughout the day, photocatalysts are often affected by the illumination direction in practical applications. This newly developed hetero-nanostructure represents the first photocatalytic system capable of high-efficiency operation under bifacial illumination, effectively triggering reactions regardless of the light's direction.
The key lies in the design of "energy band engineering." SnO2 is a wide-bandgap semiconductor, while SnS is a narrow-bandgap semiconductor. Their combination forms a "staggered-type heterojunction." When light strikes the material, the generated electrons and holes move in different directions, allowing the electrons to react with CO2 to produce CH4. This efficient charge separation mechanism dramatically boosts reaction efficiency. Furthermore, SnS extends the light absorbance range from ultraviolet to visible and even near infrared regions, allowing the material to utilize a broader solar spectrum and improve energy conversion efficiency.
Experimental results under a solar simulator show that the CO2 conversion efficiency of this new composite photocatalyst is several times higher than that of single-component materials. The NSYSU team also successfully controlled the reaction pathway by varying the lengths of the SnS nanorods, demonstrating an exceptional and selective production rate for specific fuels. Even after hours of continuous reaction, the material's structure remained stable with no significant decay in catalytic activity, demonstrating excellent long-term stability and future application potential. Compared to many photocatalysts that require expensive noble metals, the tin-based materials selected by Associate Professor Lee's team are low-cost, environmentally friendly, and have excellent photocatalytic performance, aligning with sustainable development goals while opening new possibilities for carbon recycling and renewable energy technology.
